The Food and Drug Administration (FDA) has announced it will make a pandemic era change permanent by allowing more pharmacies, including large chains and mail-order companies, to dispense pills that are commonly used in medication abortions.
The change could expand access at both brick-and-mortar stores and online pharmacies.
But this federal change won't likely have any impact on people seeking those drugs here in Ohio.
Under the rule change, women could get a prescription via telehealth consultation with a health professional, and then receive the pills through the mail, where permitted by law. That's not the case in Ohio, said Ohio Right to Life President Mike Gonidakis. He notes Ohio women seeking abortions are required, by state law, to go to a doctor or clinic in person to get a prescription for the drugs.
"Ohio requires a woman to visit, in person, with a physician prior to being administered RU 486 (Mifepristone) or other abortifacients," Gonidakis said.
Pro-Choice Ohio Deputy Director Jaime Miracle agreed this federal change won't impact Ohio women.
"Unfortunately, yes, once again politics trumps science in Ohio and will not be able to benefit directly from this FDA change," Miracle said.
Legislation that was originally passed in Ohio nearly 20 years ago prevents anyone other than a doctor from providing abortion-inducing drugs in Ohio.
The American College of Obstetricians and Gynecologists has spoken out in support of the Biden administration's policy change for abortion inducing drugs. And the Ohio Department of Health's annual abortion report shows those drugs are preferred choices among women who get abortions here during the earliest points in pregnancy. More than half of the 21,813 abortions performed in Ohio in 2021 involved the use of drugs that are affected by the FDA's policy change.
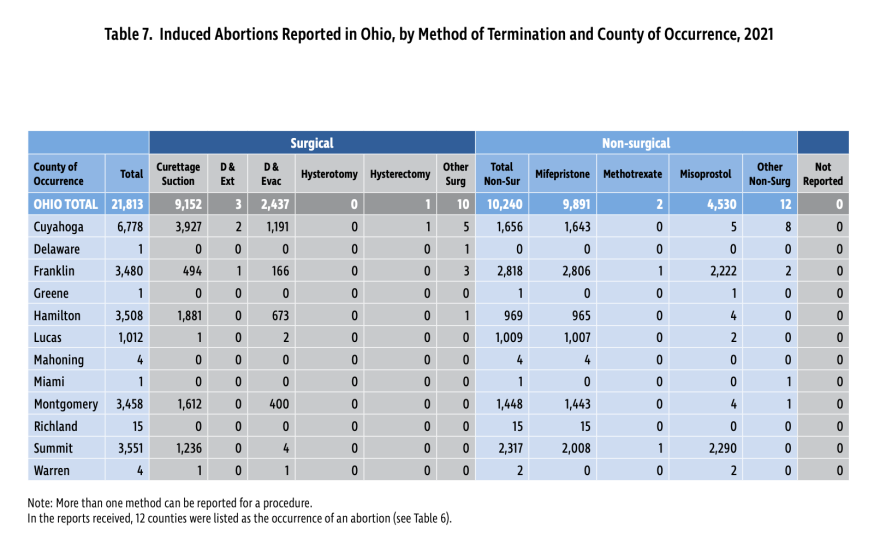
A combination of abortion inducing drugs are typically used to terminate pregnancies up to 10 weeks of gestation. Mifepristone is taken to dilate the cervix and block pregnancy hormones. Then 24 to 48 hours later, Misoprostol is taken, causing the uterus to contract and expel its contents.
The FDA reports more than 3.7 million U.S. women have used mifepristone since its approval. Bleeding is a common side effect and the agency reports serious complications from the drugs are rare.
Because of Ohio's restrictions, it's doubtful pharmacies here in the Buckeye State will carry the drug. But some pharmacies in other states might decide to stock and dispense the drugs. Amy Thibault, Lead Director of External Communications for CVS Pharmacy said in a written statement, "We’re reviewing the FDA’s updated Risk Evaluation and Mitigation Strategy (REMS) drug safety program for mifepristone to determine the requirements to dispense in states that do not restrict the dispensing of medications prescribed for elective termination of pregnancy."



